Active Pharmaceutical Ingredients
As pharmaceutical suppliers, we have developed our product range of niche, small-molecule API’s to meet the growing demands of the pharmaceutical industry. We actively research and innovate in order to bring new API’s to market in order to support our customers.
Standard API’s
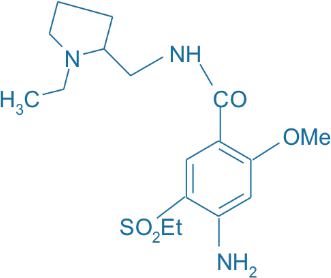
Amisulpride
Main Therapeutic Application: Antipsychotics agent / Neuroleptic agent
CAS-RN: 71675-85-9
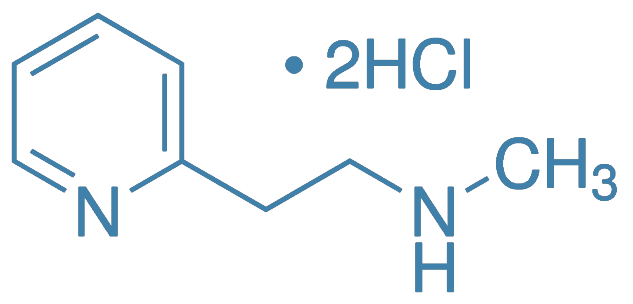
Betahistine Dihydrochloride
Main Therapeutic Application: Antivertigo agent - agonist of histamine
CAS-RN: 5579-84-0
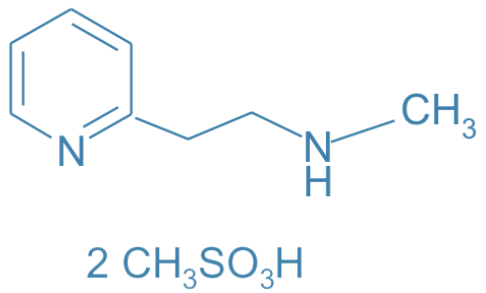
Betahistine Dimesilate
Main Therapeutic Application: Antivertigo agent - agonist of histamine
CAS-RN: 54856-23-4
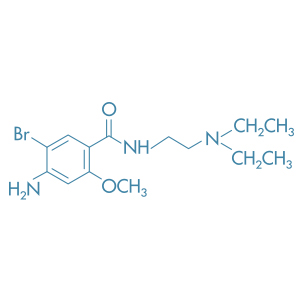
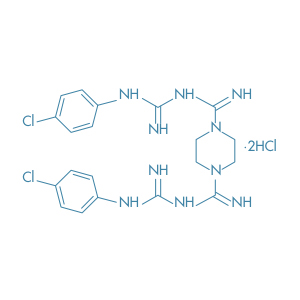
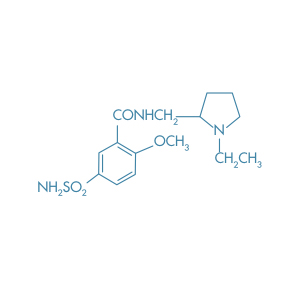
Bromopride
Main Therapeutic Application: Antiemetic agent
CAS-RN: 4093-35-0
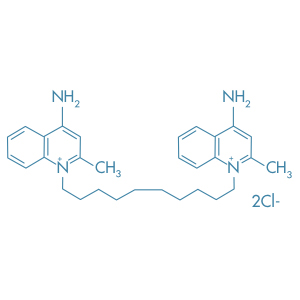
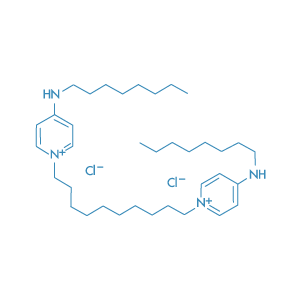
Dequalinium Chloride
Main Therapeutic Application: Antiseptic agent
CAS-RN: 522-51-0
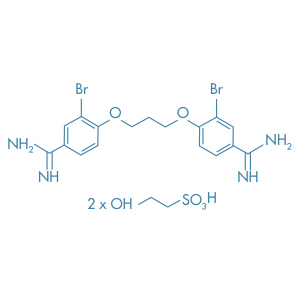
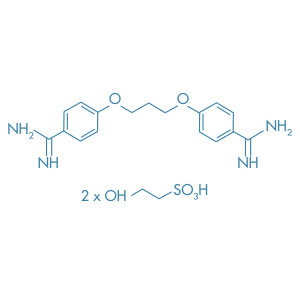
Dibrompropamidine Isetionate
Main Therapeutic Application: Disinfectant / Antiseptic
CAS-RN: 614-87-9
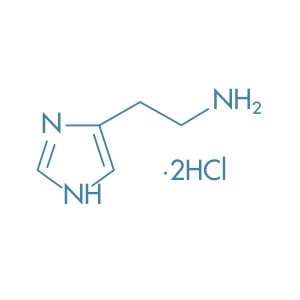
Histamine Dihydrochloride
Main Therapeutic Application: Immunostimulants / Antineoplastic agent / Diagnostic agent
CAS-RN: 56-92-8
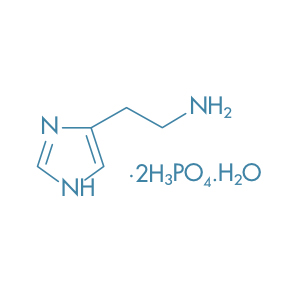
Histamine Diphosphate Monohydrate
Main Therapeutic Application: Diagnostic Agent
CAS-RN: 51-74-1
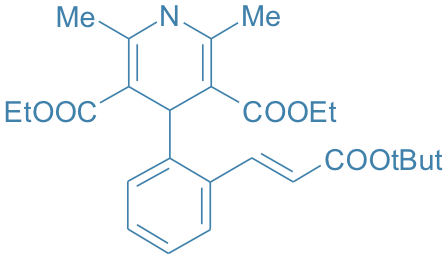
Lacidipine
Main Therapeutic Application: Antihypertensive
CAS-RN: 103890-78-4
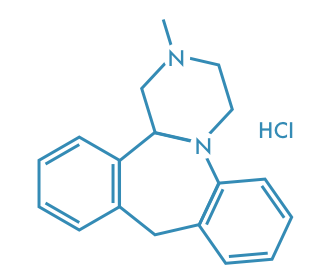
Mianserin Hydrochloride
Main Therapeutic Application: Antidepressant
CAS-RN: 21535-47-7
Octenidine Dihydrochloride
Main Therapeutic Application: Antiseptic
CAS-RN: 70775-75-6
Picloxidine Dihydrochloride
Main Therapeutic Application: Antiseptic agent
CAS-RN: 19803-62-4
Propamidine Isetionate
Main Therapeutic Application: Antiseptic agent topical
CAS-RN: 140-63-6
Sulpiride
Main Therapeutic Application: Antipsychotic agent / Neuroleptic agent
CAS-RN: 15676-16-1
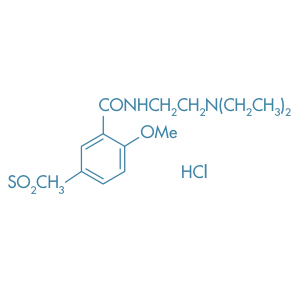
Tiapride Hydrochloride
Main Therapeutic Application: Antipsychotic agent/ Neuroleptic agent
CAS-RN: 51012-33-0
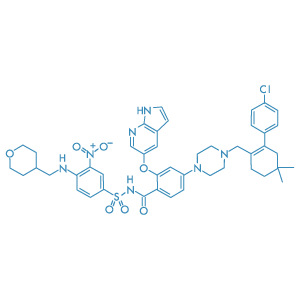
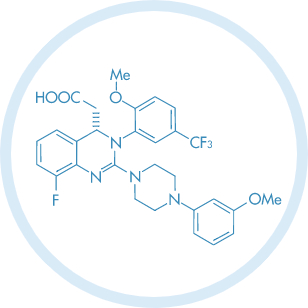
Venetoclax
Main Therapeutic Application: Antineoplasic
CAS-RN: 1257044-40-8
Get the completeAPI's porfolio?
Recieve an email with all the information about our molecules
DownloadProducts Pipeline
The API market is evolving.
The regulatory environment is more challenging than ever and being a pharmaceutical API suppliers demands more than just API production. Our clients need partners with the expertise to help them achieve their business goals at the right speed and quality level.
Our service goes beyond API manufacturing.
We build relationships based on trust and brand prestige. At LEBSA, we work every day to continually improve our processes. We also actively research and innovate in order to bring new API’s to market and support our API pharma customers.